Introduction to the world of the smallest parts
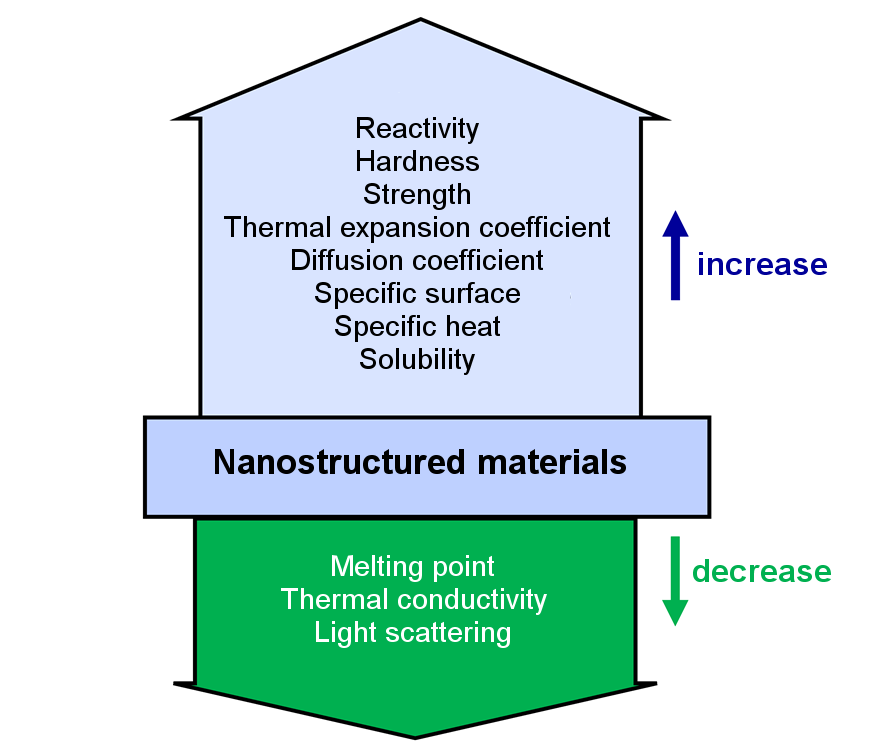
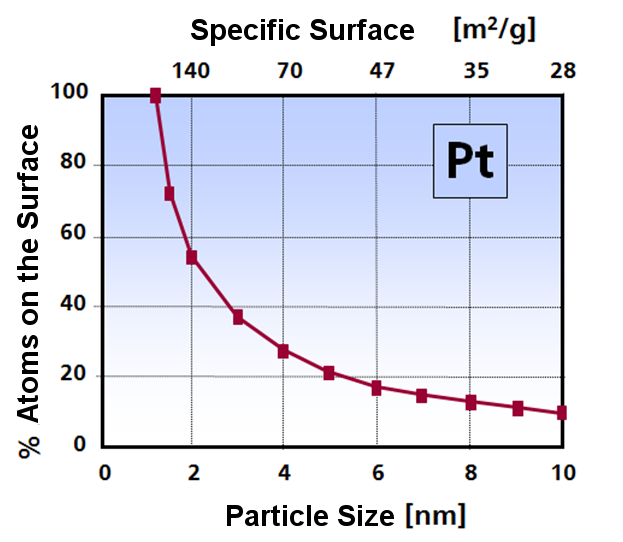
Nano is a prefix that marks the billionth part of something (10-9), comes from the Greek and literally means dwarf. So it's actually something very small, not just invisible to the eyes, but also not visible with conventional microscopes. Technology means the application of knowledge in technical processes, ie for the production of products. So what can be so special about something you almost can not perceive and how can you make products for which extreme growth rates are predicted in the future?
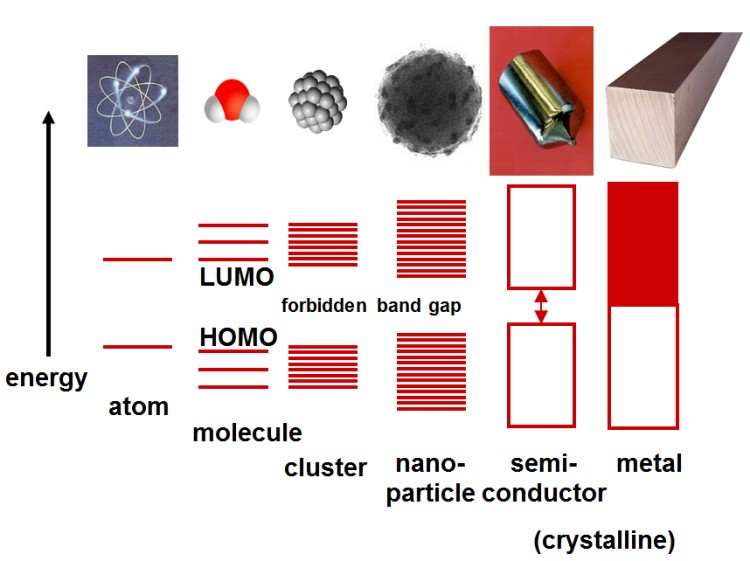
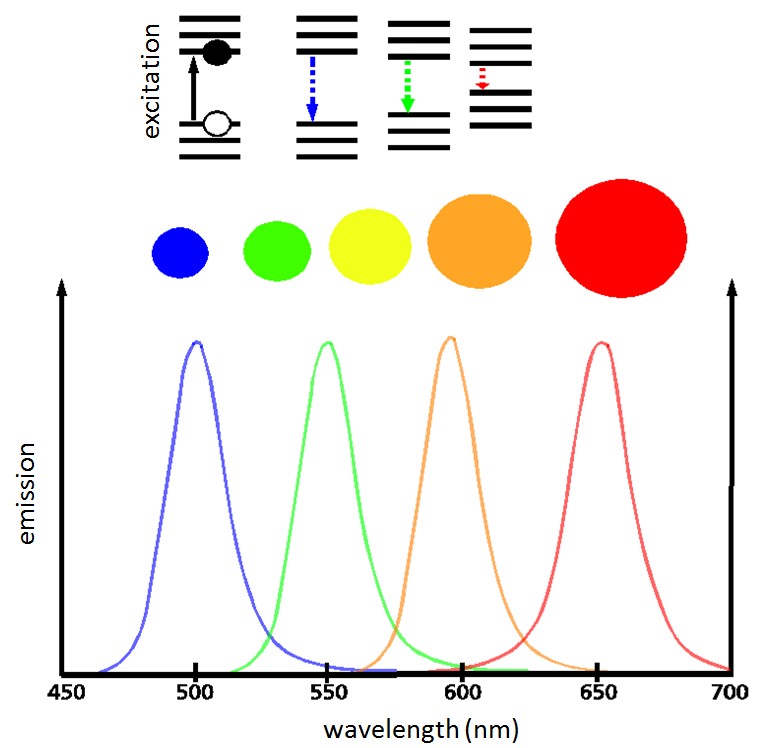
The development of nanotechnology (NT) was accelerated and by the development of scanning tunneling or scanning probe microscopy developed by IBM in the mid-80s of the last century. This makes it possible not only to make individual atoms "visible" but also to manipulate them with specially equipped finest tips.
Nanotechnology is the application of nanosciences - the study of nanoscaled systems - which can be the basis for future products and processes. These include, for example, many biological systems that are nanoscale in their functional units (eg, DNA), forming the basis of life.